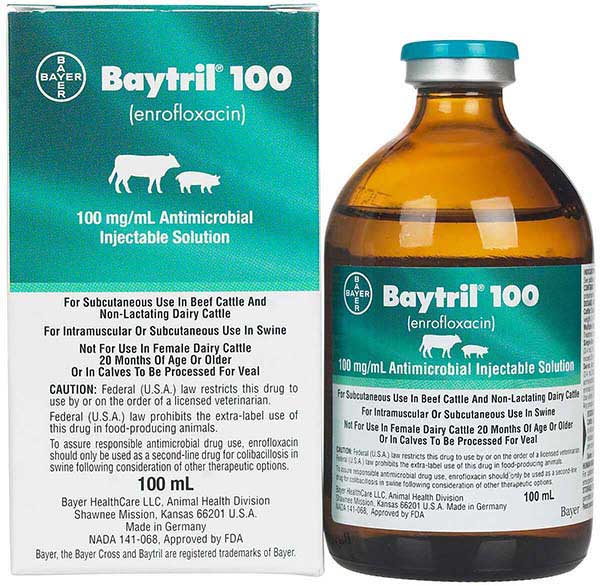
(Baytril) is an oral or injectable antimicrobial drug with a broad spectrum of action for the treatment of bacterial infections and mycoplasmosis in poultry. It is used in the treatment of bacterial diseases in chickens and poults caused by gram-positive and gram-negative bacteria and mycoplasmas sensitive to enrofloxacin. The drug is a clear sterile solution of a light yellow color. Baytril 10% (Baytril) is a drug of domestic production, not inferior in its effectiveness to foreign drugs.
Baytril10% Injectable Solution
A5581 RVM
Active Constituents:
Enrofloxacin 10% in a solution for injection. Available in 50mL bottles.
Action:
Baytril inhibits the enzyme DNA gyrase which plays a major role in the replication of the DNA. Inhibition of this enzyme leads to functional disturbances with blockage of certain stages in synthesis, resulting in death of the bacterium. This mode of action rules out cross resistance between Baytril and other anti-bacterials commonly used in veterinary medicine.
Indication:
Baytril 10% is reserved for use in New Zealand for the following indications only:
Bacterial infections of bones in pigs and cattle
Mastitis in cows where the causative organisms have been established to be either E.coli or Pseudomonas strains sensitive to enrofloxacin. Appropriate intramammary treatment with another antibiotic can be used in combination with systemic Baytril treatment.
Infection of the (male) reproductive tract (prostatitis, vesiculitis, orchitis) in the bull.
Certain infections of cattle and pigs in locations where poor tissue penetration by other antimicrobial drugs can be expected and where the condition is caused by a susceptible organism which does not respond readily to other antibiotics.
Indiscriminate use of antibiotics may lead to the development of resistance. Baytril should not be used for routine preventive purposes and should be used in individual animals in line with the above recommendations. Treatment of groups of food producing animals is only recommended when culture and sensitivity testing indicate Baytril is the only product likely to be effective. Susceptibility testing is recommended where feasible.
Dosage:
For s/c injection in cattle and i/m injection in pigs.
CATTLE and PIGS: Standard dose rate 2.5mL/100kg b.w/day for 3 days.
Severe respiratory infections: 5.0mL/100kg b.w/day for 3 days.
Withholding Times:
MEAT: Cattle and pigs producing meat and offal for human consumption must not be sold for slaughter either during treatment or within 7 days of the last treatment. MILK: Milk intended for sale for human consumption must be discarded during treatment and for 7 days, 14 milkings (168 hours) following the last treatment.https://kihorsemed.com/


Reviews
There are no reviews yet.